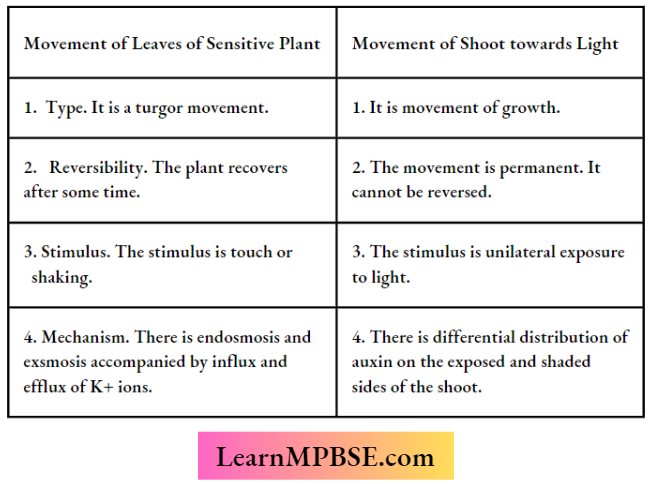
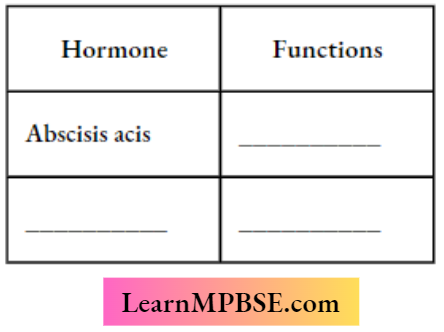
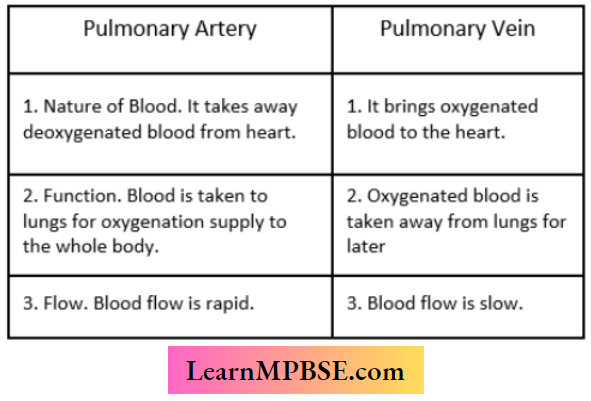
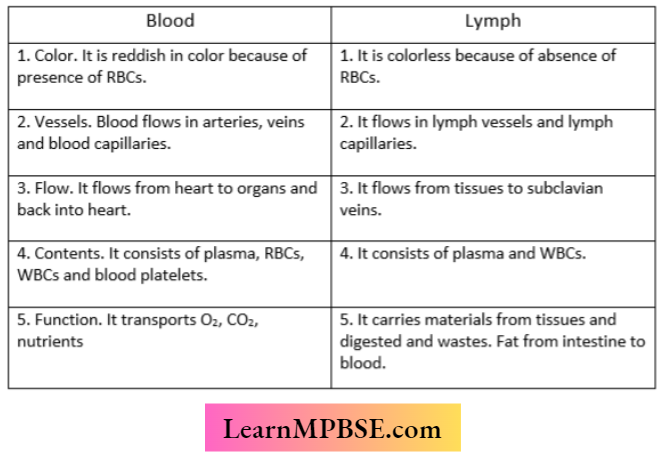
MP Board Class 7 Maths Solutions For Chapter 2 Fractions and Decimals
1. \( 5 \times 2 \frac{3}{7}\)
Solution: \( 5 \times 2 \frac{3}{7}=5 \times 12 \frac{1}{7}=\frac{85}{7}=12 \frac{1}{7}\)
2. \(1 \frac{4}{9} \times 6\)
Solution: \( 1 \frac{4}{9} \times 6=a \times 6=\frac{78}{9}=8 \frac{6}{9}\)
what is
1. \( \frac{1}{2}\) of 10 ?
Solution:
\( \frac{1}{2} \text { of } 10=\frac{1}{2} \times 10=\frac{10}{2}=5\)2. \( \frac{1}{4}\) of 16 ?
Solution:
\( \frac{1}{4} \text { of } 16=\frac{1}{4} \times 16=\frac{16}{4}=4\)3. \( \frac{2}{5}\)
Solution: \( \frac{2}{5} \text { of } 25=\frac{2}{5} \times 25=\frac{50}{5}=10\)
Fill in these boxes:
1) \( \frac{1}{2} \times \frac{1}{7}=\frac{1 \times 1}{2 \times 7}=\frac{1}{14} \)
2) \( \frac{1}{5} \times \frac{1}{7}=\frac{1 \times 1}{5 \times 7}=\frac{1}{35} \)
3) \( \frac{1}{7} \times \frac{1}{2}=\frac{1 \times 1}{7 \times 2}=\frac{1}{14} \)
4) \( \frac{1}{7} \times \frac{1}{5}=\frac{1 \times 1}{7 \times 5}=\frac{1}{35} \)
Mp Board Class 7 Book Solutions
Find:
1) \( \frac{1}{3} \times \frac{4}{5} \)
Solution: \( \frac{1}{3} \times \frac{4}{5}=\frac{1 \times 4}{3 \times 5}=\frac{4}{15} \)
2) \( \frac{2}{3} \times \frac{1}{5} \)
Solution: \( \frac{2}{3} \times \frac{1}{5}=\frac{2 \times 1}{3 \times 5}=\frac{2}{15} \)
Solutions To Try These
1) \( \frac{8}{3} \times \frac{4}{7} \)
Solution: \( \frac{8}{3} \times \frac{4}{7}=\frac{8 \times 4}{3 \times 7}=\frac{32}{21} \)
2) \( \frac{3}{4} \times \frac{2}{3} \)
Solution: \( \frac{3}{4} \times \frac{2}{3}=\frac{3 \times 2}{4 \times 3}=\frac{6}{12}=\frac{6 \div 6}{12 \div 6}=\frac{1}{2} \)
MP Board Class 7 Maths Solutions For Chapter 2 Fractions and Decimals
1. Find
(1) \( \frac{1}{4} of \) 1) \frac{1}{4} 2) \( \frac{3}{5} \) 3) \( \frac{4}{3}\)
1) \( \frac{1}{4} \text { of } \frac{1}{4}=\frac{1}{4} \times \frac{1}{4}=\frac{1 \times 1}{4 \times 4}=\frac{1}{16} \)
2). \( \frac{1}{4} \text { of } \frac{3}{5}=\frac{1}{4} \times \frac{3}{5}=\frac{1 \times 3}{4 \times 5}=\frac{3}{20} \)
3) \( \frac{1}{4} \text { of } \frac{4}{3}=\frac{1}{4} \times \frac{4}{3}=\frac{1 \times 4}{4 \times 3}=\frac{4}{12} \)
\( =\frac{4 \div 4}{12 \div 4}=\frac{1}{3} \)Mp Board Class 7 Book Solutions
2) \( \frac{1}{7} \) of 1) \( \frac{2}{9} \) 2) \( \frac{6}{5} \) 3) \( \frac{3}{10} \)
Solution:
1) \( \frac{1}{7} \text { of } \frac{2}{9}=\frac{1}{7} \times \frac{2}{9}=\frac{1 \times 2}{7 \times 9}=\frac{2}{63} \)
2)\( \frac{1}{7} \text { of } \frac{6}{5}=\frac{1}{7} \times \frac{6}{5}=\frac{1 \times 6}{7 \times 5}=\frac{6}{35} \)
3)\( \frac{1}{7} \text { of } \frac{3}{10}=\frac{1}{7} \times \frac{3}{10}=\frac{1 \times 3}{7 \times 10}=\frac{3}{70} \)
2. Multiply and reduce to lowest form (if possible):
1. \( \frac{2}{3} \times 2 \frac{2}{3} \)
Solution: \( \frac{2}{3} \times 2 \frac{2}{3}=\frac{2}{3} \times \frac{8}{3}=\frac{2 \times 8}{3 \times 3}=\frac{16}{9}=1 \frac{7}{9} \)
2. \( \frac{2}{7} \times \frac{7}{9} \)
Solution:
\( \frac{2}{7} \times \frac{7}{9}=\frac{2 \times 7}{7 \times 9}=\frac{14}{63}=\frac{14 \div 7}{63 \div 7}=\frac{2}{9} \)3. \( \frac{3}{8} \times \frac{6}{4} \)
Solution:
\( \frac{3}{8} \times \frac{6}{4}=\frac{3 \times 6}{8 \times 4}=\frac{18}{32}=\frac{18 \div 2}{32 \div 2}=\frac{9}{16} \)Mp Board Class 7 Maths Solutions
4. \( \frac{9}{5} \times \frac{3}{5} \)
Solution:
\( \frac{9}{5} \times \frac{3}{5}=\frac{9 \times 3}{5 \times 5}=\frac{27}{25}=1 \frac{2}{25} \)5. \( \frac{1}{3} \times \frac{15}{8} \)
Solution:
\( \frac{1}{3} \times \frac{15}{8}=\frac{1 \times 15}{3 \times 8}=\frac{15}{24}=\frac{15 \div 3}{24 \div 3}=\frac{5}{8} \)6. \( \frac{11}{2} \times \frac{3}{10} \)
Solution: \( \frac{11}{2} \times \frac{3}{10}=\frac{11 \times 3}{2 \times 10}=\frac{33}{20}=1 \frac{13}{20} \)
7. \( \frac{4}{5} \times \frac{12}{7} \)
Solution: \(\frac{4}{5} \times \frac{12}{7}=\frac{4 \times 12}{5 \times 7}=\frac{48}{35}=1 \frac{13}{35} \)
3. Multiply the following fractions:
1) \( \frac{2}{5} \times 5 \frac{1}{4} \)
Solution: \( \frac{2}{5} \times 5 \frac{1}{4}=\frac{2}{5} \times \frac{21}{4} \)
\(=\frac{2 \times 21}{5 \times 4}=\frac{42}{20}=\frac{42 \div 2}{20 \div 2} \) \( =\frac{21}{10}=2 \frac{1}{10} \)Class 7 Maths Chapter 2 Solutions Mp Board
2) \( 6 \frac{2}{5} \times \frac{7}{9} \)
Solution: \( 6 \frac{2}{5} \times \frac{7}{9}=\frac{32}{5} \times \frac{7}{9} \)
\( =\frac{32 \times 7}{5 \times 9}=\frac{224}{45}=4 \frac{44}{45} \)3) \( \frac{3}{2} \times 5 \frac{1}{3} \)
Solution: \( \frac{3}{2} \times 5 \frac{1}{3}=\frac{3}{2} \times \frac{16}{3}=\frac{3 \times 16}{2 \times 3}=\frac{48}{6} \)
\( =\frac{48 \div 6}{6 \div 6}=\frac{8}{1}=8 \)4. \( \frac{5}{6} \times 2 \frac{3}{7} \)
Solution: \( \frac{5}{6} \times 2 \frac{3}{7}=\frac{5}{6} \times \frac{17}{7}=\frac{5 \times 17}{6 \times 7}=\frac{85}{42}=2 \frac{1}{42} \)
5. \( 3 \frac{2}{5} \times \frac{4}{7} \)
Solution: \( 3 \frac{2}{5} \times \frac{4}{7}=\frac{17}{5} \times \frac{4}{7}=\frac{17 \times 4}{5 \times 7}=\frac{68}{35}=1 \frac{33}{35} \)
6. \( 2 \frac{3}{5} \times 3 \)
Solution: \( 2 \frac{3}{5} \times 3=\frac{13}{5} \times 3=\frac{13 \times 3}{5}=\frac{39}{5}=7 \frac{4}{5} \)
7.\( 3 \frac{4}{7} \times \frac{3}{5} \)
Solution: \( =\frac{75 \div 5}{35 \div 5}=\frac{15}{7}=2 \frac{1}{7} \)
\( =\frac{75 \div 5}{35 \div 5}=\frac{15}{7}=2 \frac{1}{7} \)Mp Board Maths Chapter 2 Solutions
4. Which is greater:
1) \( \frac{2}{7} \) of \( \frac{3}{4} \) or \( \frac{3}{5} \) of \( \frac{5}{8} \)
Solution: \( \frac{2}{7} \text { of } \frac{3}{4}=\frac{2}{7} \times \frac{3}{4}=\frac{2 \times 3}{7 \times 4} \)
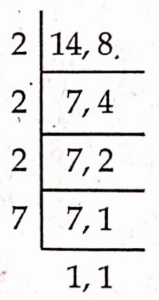
\( =\frac{6}{28}=\frac{6 \div 2}{28 \div 2}=\frac{3}{14} \) \( \frac{3}{5} \text { of } \frac{5}{8}=\frac{3}{5} \times \frac{5}{8}=\frac{3 \times 5}{5 \times 8}=\frac{15}{40}=\frac{15 \div 5}{40 \div 5}=\frac{3}{8} \)LCM = 2 x 7X 4 = 56
LCM of 14, 8 is 56
\( \frac{3}{14}=\frac{3 \times 4}{14 \times 4}=\frac{12}{56} \) \( \frac{3}{8}=\frac{3 \times 7}{8 \times 7}=\frac{21}{56} \)∴ 21 > 12
Mp Board Maths Chapter 2 Solutions
2) \( \frac{1}{2} \text { of } \frac{6}{7} \text { or } \frac{2}{3} \text { of } \frac{3}{7} \)
Solution:
\( \begin{aligned}&\begin{aligned}
\frac{1}{2} \text { of } \frac{6}{7} & =\frac{1}{2} \times \frac{6}{7}=\frac{1 \times 6}{2 \times 7}=\frac{6}{14} \\
& =\frac{6 \div 2}{14 \div 2}=\frac{3}{7}
\end{aligned}\\
&\begin{aligned}
\frac{2}{3} \text { of } \frac{3}{7}=\frac{2}{3} \times \frac{3}{7}=\frac{2 \times 3}{3 \times 7} & =\frac{6}{21} \\
& =\frac{6 \div 3}{21 \div 3}=\frac{2}{7}
\end{aligned}
\end{aligned} \)
3 > 2;
\( \frac{3}{7}>\frac{2}{7} \) \( \frac{1}{2} \text { of } \frac{6}{7}>\frac{2}{3} \text { of } \frac{3}{7} \)5. Saili plants 4 saplings,in a row,in her garden. The distance between two adjacent saplings is \( \frac{3}{4} \) m. Find the distance between the first and the last sapling.
Solution:
Distance between two saplings = \( \frac{3}{4} \) m.
Distance between the first and the last sapling = \( 3 \times \frac{3}{4}=\frac{3 \times 3}{4}=\frac{9}{4}=2 \frac{1}{4} \mathrm{~m} \)
6. Lipika reads a book for \( 1 \frac{3}{4} \) hours everyday. She reads the entire book in 6 days. How many hours in all were required by her to read the book ?
Solution:
No. of hours taken by Lipika to read the bookin a day = \( 1 \frac{3}{4} \)
No. of days taken toread the entire book = 6 days
Time takenby Lipika to read the book
\( =1 \frac{3}{4} \times 6 \) \( \begin{aligned}& =\frac{(1 \times 4+3)}{4} \times 6=\frac{7}{4} \times 6=\frac{42}{4} \\
& =\frac{42 \div 2}{4 \div 2}=\frac{21}{2}=10 \frac{1}{2} \text { hours }
\end{aligned} \)
7. A car covers 16 km using 1 litre of petrol.Howmuch distance willit cover using \( 2 \frac{3}{4} \) litres of petrol ?
Solution:
Distance covered with 1 litre of petrol = 16 km
Distance covered with \( 2 \frac{3}{4} \) litres of petrol = \( 16 \times 2 \frac{3}{4} \)
\( \begin{aligned}& =16 \times\left(\frac{2 \times 4+3}{4}\right)=16 \times \frac{11}{4} \\
& =\frac{16 \times 11}{4}=\frac{176}{4}=44 \mathrm{~km}
\end{aligned} \)
Class 7 Mp Board Maths Question Answers
8.
1) 1) Provide the number in the box such that □, \( \frac{2}{3} \) × □ = \( \frac{10}{30} \)
2) The simplest form of the number obtained in □ is…………….
Solution:
1. \( \frac{2}{3} \) x = \( \frac{5}{10} \) \(\frac{10}{30} \)
2. \( \frac{5}{10}=\frac{5 \div 5}{10 \div 5}=\frac{1}{2} \)
2) 1) Provide the number in the box □, such that \( \frac{3}{5}\) x □ = \( \frac{24}{75}\)
2) The simplest form of the number obtained in □ is ……….
Solution:
1) \( \frac{3}{5} \times \frac{8}{15}=\frac{24}{75} \)
2) \( \frac{8}{15} \)
Think, Discuss And Write
1) Will the reciprocal of a proper fraction be again a proper fraction?
Solution. No, the reciprocal of a proper fraction will not be a proper fraction. It will be an improper fraction.
Example: \( \frac{2}{9} \text { its reciprocal is } \frac{9}{2} \)
\( \frac{2}{9} \) is aproper fraction; \( \frac{9}{2} \) is animproper fraction.
2) Will the reciprocal of an improper fraction be again animproper fraction?
Solution: No, the reciprocal of an improper fraction will not.be an improper fraction again.It will be a proper fraction.
Example: \( \frac{5}{4} \) is animproper fraction.But its reciprocal \( \frac{4}{5} \) is a proper fraction.